By Elizabeth Edmiston
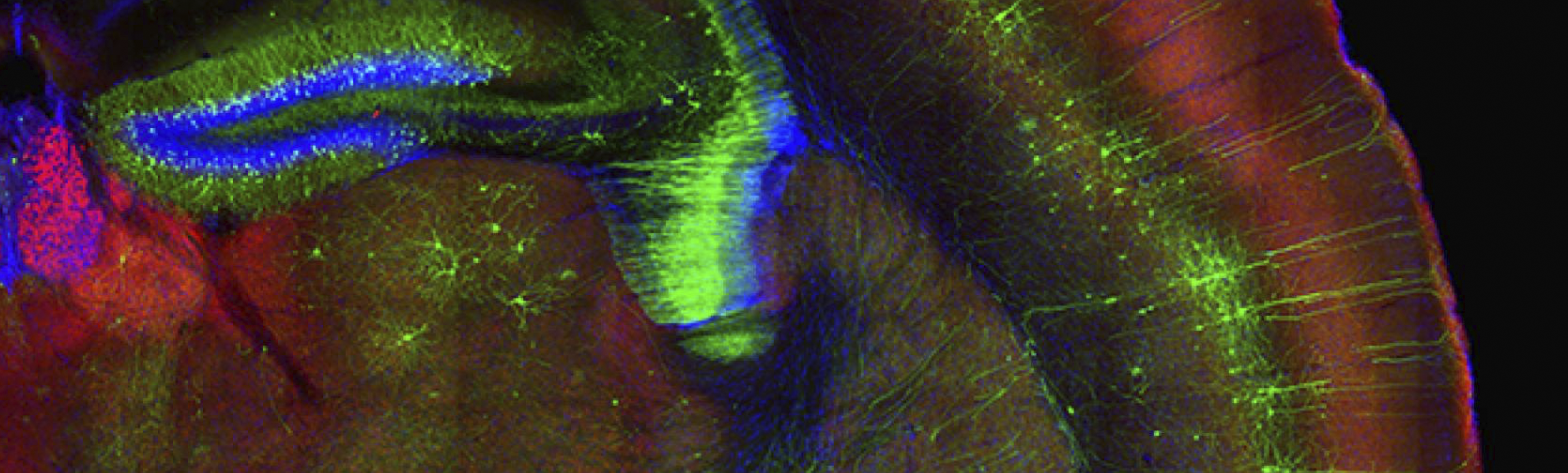
By mid-century, the number of people suffering from neurodegenerative diseases is expected to double1. With the recent string of late stage clinical trial failures in Alzheimer’s Disease, it is still an open question as to whether treatments can be developed for a disease that is definitively diagnosed post-mortem. “We have come to realize there is no one single target or magic bullet to stop the neurodegenerative process,” said Dr. Eric Huang, MD/PhD, a professor of Pathology and member of ImmunoX at UCSF. “By the time patients have cognitive impairment, it is probably past the point of no return.” In other words, scientists are coming to realize that the hope for new treatments capable of changing the course of disease lies in developing innovative approaches that deepen our understanding of the early biological underpinnings of neurodegeneration. Innovation Ventures interviewed three UCSF investigators doing just that.
Dr. Huang’s research focuses on the pathogenesis of familial frontotemporal dementia (FTD) caused by a dominant mutation in the progranulin (GRN) gene. His group has developed several GRN-mutated FTD mouse models and employs single cell transcriptome analysis in these models to track transcriptional changes in key cell types – neurons, microglia, and astroglia – over the course of the disease.
“We focus on the onset and contributions of glial pathology in neurodegeneration,” said Dr. Huang. “Under normal physiological conditions there is a very tight homeostasis between glia and neurons…they are very closely linked at the functional and structural level. What we are finding from single cell transcriptomic data, collected from mutant mice at a very young age to disease end stage, is disease progression is really a disruption of the homeostatic state between neurons and glia.”
Furthermore, through campus collaborations, the Huang lab is able to use this approach to analyze human tissue from FTD patients as well as Alzheimer’s patients, and correlate transcriptomic findings with peripheral inflammatory markers. This allows them to bridge the gap between their findings in animal models and disease outcomes in humans. It also permits them to discover disease biomarkers and compare sub-types of neurodegeneration to reveal similar and divergent mechanisms of disease. His team hopes their molecular profiling approach will enable a better understanding of the cells and pathways involved in initiating disease, provide the ability to detect and diagnose disease before cognitive symptoms arise, and offer insight into preventative measures to restore cellular homeostasis in the brain.
Assistant professor in the Department of Biochemistry and Biophysics and member of the Institute for Neurodegenerative Diseases, Martin Kampmann believes that “we are still at the very early stages of understanding the molecular and cellular processes that drive [neurodegeneration]. What we are really lacking is an understanding of the mechanism by which certain gene variants cause disease or increase disease risk, and what we can do about that therapeutically.”
To address this knowledge gap, his group is combining CRISPR-based functional genomics and induced pluripotent stem cell (iPSC)-derived neurons and glial cells to conduct genetic screens and directly study molecular and cellular processes in human cells.
Dr. Kampmann’s lab has developed CRISPR-based technologies that can knock-down or overexpress genes and they have generated large libraries of guide RNAs, allowing them to query almost every gene in the genome. They are also systematically studying genetic interactions by perturbing combinations of genes and measuring the resulting outcomes in a panel of high through-put assays. With this rather industrial approach, his lab can better model the complex genetic backgrounds observed in the human population. The next step in their work is to generate organoids and 3D models of disease, which better recapitulate the way different brain cells interact. The Kampmann lab foresees this platform can be used to elucidate new therapeutic targets, which could become the basis for drug development, aid in identifying patients that will respond to a therapeutic candidate and uncover the relevant mechanism of action of new compounds.
Assistant professor in the departments of Physical Therapy and Rehabilitation Science, and Radiology and Biomedical Imaging, Dr. Myriam Chaumeil focuses on developing new MRI-based imaging methods to understand brain metabolism in disease conditions. Specifically, her lab is trying to see how the brain works and metabolizes different substrates and how that is affected in disease.
“Alzheimer’s disease is interesting, we know metabolism is affected, and we know patients have early changes in glucose and sugar consumption, but we don’t know how it is linked to the disease course,” said Dr. Chaumeil. “If we can find which metabolic pathways are affected, we can then design drugs to target those pathways and hopefully prevent the disease from happening or improve life expectancy and quality.”
Hyperpolarized 13C MR Spectroscopic Imaging is comparable to PET imaging, but is advantageous as it is non-radioactive, more cost-effective, and the acquisition time is very short – to acquire an image takes less than a minute. Dr. Chaumeil imagines a future where it could be added to the end of any clinical exam to glean more information about brain function and metabolism, which in turn may improve diagnosis, prognosis, and therapeutic monitoring. Her group is advancing this technology by improving the acquisition schemes as well as designing new probes that can cross the blood-brain barrier and target specific disease pathways.
Despite the challenges associated with neurodegenerative disease research, it has never been a more exciting time in the field. Improvements in advanced genetic tools, imaging and screening technologies, and model systems are allowing for discoveries previously unachievable. UCSF prides itself on its faculty and the breadth of approaches they are taking to address the most pressing health issues of our time. The truly collaborative nature of our organization allows us to attract and keep world-class faculty conducting the most cutting-edge research to accelerate the pace of discovery. Dr. Chaumeil perhaps summed it up best when she said, “It is a highly collaborative place…all of the technologies are available, and they are all state-of-the-art. If you have an idea, any hypothesis, you can test it. There is no barrier to what you can try. Scientifically speaking, you can do anything, as long as it is ethical.”
...
